Practitioner Research Review – May 2018
Dr. Michael Ruscio’s Monthly – Future of Functional Medicine Review Clinical Newsletter
Practical Solutions for Practitioners
In Today’s Issue
Research
- Low FODMAP in 2017: Lessons Learned From Clinical Trials and Mechanistic Studies
- Effect of a Preparation of Four Probiotics on Symptoms of Patients with Irritable Bowel Syndrome: Association with Intestinal Bacterial Overgrowth
- 25-Hydroxyvitamin-D3 Serum Modulation After Use of Sunbeds Compliant with European Union Standards: A Randomized Open Observational Controlled Trial
- Rapid-Fire Research – Ultra Concise Summaries Of Noteworthy Studies
- Follow-Up of Patients with Functional Bowel Symptoms Treated with a Low FODMAP Diet
- Aberrant Motility in Unaffected Small Bowel is Linked to Inflammatory Burden and Patient Symptoms in Crohn’s Disease
- A Proof-of-Concept Study Showing Antibiotics to be More Effective in Irritable Bowel Syndrome With Than Without Small Intestinal Bacterial Overgrowth: A Randomized, Double-Blind, Placebo-Controlled Trial
- Prevalence of Thyroid Dysfunction and Autoimmunity in the Older Population and Implications of Age-Specific Reference Ranges
- Subclinical Thyroid Dysfunction and the Risk of Cognitive Decline: A Meta-Analysis of Prospective Cohort Studies
- Factors Affecting Gastrointestinal Absorption of Levothyroxine: A Review
![]()
Research
*Please note: the case study and research studies are not meant to be mutually reinforcing. There is often concept overlap, however the research studies are a collection of the most clinically meaningful research that has been published recently.
Low FODMAP in 2017: Lessons Learned From Clinical Trials and Mechanistic Studies
https://www.ncbi.nlm.nih.gov/pubmed/28345807
Study purpose
- Review of mechanistic and clinical trial findings of the low FODMAP diet.
Intervention:
- Review, non-systematic
Main Results:
Mechanisms
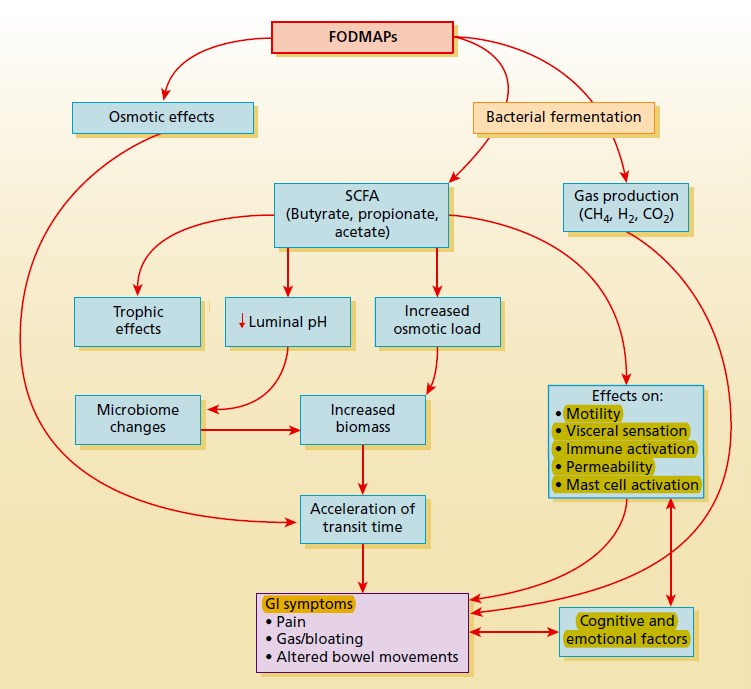
- Emerging evidence suggests hypersensitivity to gas pressure, rather than overproduction of gas, may drive a subset of FODMAP induced symptoms.
- This indicates that colonic hypersensitivity, rather than greater gas production or distension, drives FODMAP- related symptoms in some IBS patients
- The low FODMAP may also lead to clinical improvements via a reduction in histamine.
- Histamine, a signaling molecule known to underlie IBS symptoms, may also be affected by the low FODMAP diet. McIntosh et al. [21] compared urinary metabolomic profiles of 40 IBS patients after 21 days of a low- or high- FODMAP diet. Following dietary intervention there was a significant separation in urinary metabolomic profiles of patients with IBS in the two diet groups. In the low FODMAP diet group, urinary histamine level decreased significantly after the intervention (P<.05) compared to the high-FODMAP group.
- Speculatively, a low FODMAP diet may stabilize mast cells. SCFA or gas pressure may activate mast cells.
- The authors postulate that degranulation of mast cells may occur due to direct signaling from short chain fatty acids (SCFAs) or from intestinal distension via fermentation, thereby modulating IBS symptoms.
- The effect on the microbiota and on SCFAs have been inconsistent.
- Demonstrated a reduction in the proportion and concentration of Bifidobacteria. Another study did not demonstrate a decrease in Bifidobacteria but did show a decrease in total bacteria abundance
- Halmos et al. found no change in SFCA concentration between the low FODMAP diet and a habitual Australian diet, while others have observed a decrease in SCFA compared to a habitual diet.
- A low FODMAP diet may reduce inflammation (cytokines).
- Levels of IL-6 and Il-8 (but not TNF-α) both decreased significantly after 3 weeks of a low FODMAP diet
- Levels of IL-6 and Il-8 (but not TNF-α) both decreased significantly after 3 weeks of a low FODMAP diet
Clinical Outcomes
- RCTs have shown improvement in 52-76% of IBS patients.
- Subsequent retrospective and randomized studies of dietary FODMAP restriction have reported symptomatic improvement in 52%–76% of IBS patients.
- However, some comparative trials have found the low FODMAP diet to be no better than more traditional dietary advice.
- The results of randomized controlled trials for the low FODMAP diet in IBS patients in IBS patients have not been uniformly positive, especially when compared with active interventions in a more “real world” setting where food was not supplied to subjects.
- Bohn et al. compared the low FODMAP diet to standard dietary advice and found that about half of each group improved with the intervention, with no significant difference between the two groups after 4 weeks. Each group received dietitian counseling, and all IBS subtypes were included. Similar improvements in each group were noted for most individual symptoms as well (bloating, abdominal pain).
- Our group recently completed the first US comparative effectiveness trial comparing the low FODMAP diet vs usual dietary recommendations in IBS patients with diarrhea (IBS-D) using a similar study design in 92 patients. There was no significant difference between the interventions for the primary endpoint of adequate relief (52% with a low FODMAP diet vs 41% with usual dietary recommendations).
- Does this mean a low FODMAP diet isn’t worth considering? No. Important note: the low FODMAP diet is not the only diet that works for IBS. Standard dietary advice can help. However, there appears to be a great number of symptoms improved from the low FODMAP diet. So, it’s reasonable to say, start with dietary basics, then consider low FODMAP if there is a non-optimal response to standard dietary advice. Just as the British Dietetic Association has concluded in their IBS practice guidelines. (1)
- However, a significantly greater proportion in low FODMAP diet group than the usual dietary recommendation group experienced improvement in abdominal pain and bloating, two of the most bothersome complaints associated with IBS. In addition, significant improvements were seen in stool consistency, stool frequency, and urgency compared to usual dietary recommendations for IBS. Significant improvements in quality of life measures, as well as anxiety, were seen in the low FODMAP diet compared to usual dietary recommendations for IBS.
Additional Results:
- “Undigested, non-absorbed FODMAPs create an osmotic load and are then fermented by small intestinal and colonic bacteria. This leads to the production of short-chain fatty acids and gases (hydrogen, methane, carbon dioxide), which can trigger symptoms particularly in patients who have underlying abnormalities in gut motility and visceral sensation. Collectively, these effects can exert primary and secondary effects on motility, visceral sensation, and the gut microbiota that may result in symptoms of cramping, bloating, distention, and flatulence in a subset of IBS patients.”
Authors Conclusion:
- “The efficacy of a low FODMAP diet for IBS is becoming increasingly obvious but several areas remain to be clarified: (i) the mechanism(s) by which FODMAP restriction improves symptoms, (ii) long-term effects/safety in terms of gut microbiota and potential nutritional deficiencies, (iii) standardization of a reintroduction protocol.”
Clinical Takeaways:
- The low FODMAP diet is efficacious for IBS, however, it may be more practical to start with easier to implement standard dietary recommendations first and consider the low FODMAP as a secondary dietary intervention.
- The low FODMAP diet may lead to symptomatic improvement via a variety of mechanisms, including:
- A reduction of gas and gas pressure
- A reduction in certain bacterial populations (this has not been conclusively shown)
- Reduced immune activation. More specifically, a reduction in histamine & mast cell stabilization – potentially as a byproduct of reduced SCFA and gas pressure
- Elimination should be followed by later reintroduction to tolerance
Dr. Ruscio Comments
Low FODMAP diets are a prime consideration for those with IBS, however, this study makes a good point to not forget about the simple recommendations laid out via standard dietary guidelines. Said more simply, if someone has done nothing to improve their diet yet, simple changes should be employed first. Many clinicians will see patients who have already made these changes and a low FODMAP diet could be initiated right away.
Also, remember not to think of the gut in a reductionistic fashion. Currently, there is a tendency to reduce gut health to the view of microbiota-associated changes. The microbiota is one important aspect, but it is crucial not to overlook clinical outcomes (improved symptoms) and immune system changes (reduction in cytokines, histamine and potential mast cell stabilization).
Effect of a Preparation of Four Probiotics on Symptoms of Patients with Irritable Bowel Syndrome: Association with Intestinal Bacterial Overgrowth
https://www.ncbi.nlm.nih.gov/pubmed/29508268
Study purpose
- To assess the impact of probiotics on IBS and determine the impact of associated SIBO
Intervention:
- 5 patients with IBS + SIBO and 21 patients with IBS without SIBO.
- Prospectively administered Saccharomyces boulardii, Bifidobacterium lactis, Lactobacillus acidophilus, and Lactobacillus plantarum (Lactolevure®) every 12 h for 30 days.
- This would essentially equal a combination of category 1 and category 2 probiotics using my classification system.
- SIBO assessed via duodenal culture. IBS defined via Rome III criteria.
Main Results:
- In short: probiotics worked better for those with IBS + SIBO than IBS without SIBO.
- Thirty days after the end of treatment the following was found:
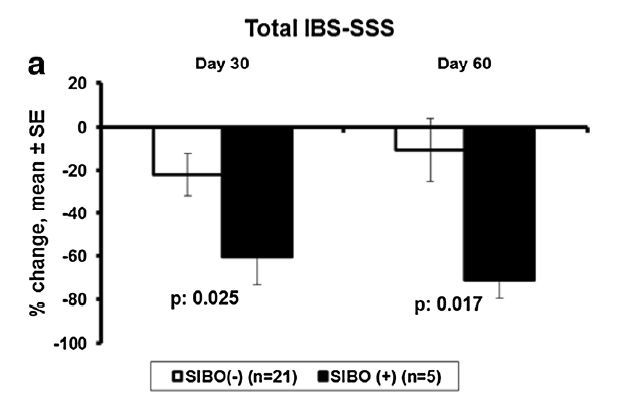
- IBS + SIBO group
- 71.3% decrease in the total IBS score
- IBS without SIBO group
- 10.6% decrease in the total IBS score
- Probiotics improved constipation type IBS irrespective of SIBO presence
- Our data show a beneficial effect of the intake of probiotics also for IBS-C patients, irrespective of the presence of SIBO
- IBS + SIBO group
- The benefit of probiotics was greater among patients with a proinflammatory cytokine pattern in the duodenal fluid.
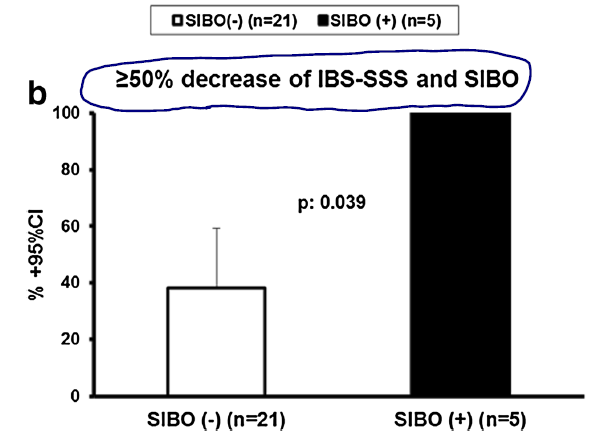
- Ten patients were pro-inflammatory based on their cytokine patterns in the duodenal fluid and 16 were normal; any ≥ 50% decrease of the IBS-SSS in visit 3 was achieved in seven (70%) and six (37.5%), respectively.
- Ten patients were pro-inflammatory based on their cytokine patterns in the duodenal fluid and 16 were normal; any ≥ 50% decrease of the IBS-SSS in visit 3 was achieved in seven (70%) and six (37.5%), respectively.
Additional Results:
- “All patients with SIBO presented with ≥ 50% decrease of the total IBS-SSS on visit 3 compared to 38.1% of patients without SIBO”
- “Our findings point towards an anti-inflammatory mechanism of action of probiotics since most of the efficacy was shown for patients with a pro-inflammatory cytokine pattern in the duodenal fluid. This insight into the mechanism of probiotic action in IBS has never been shown before.”
Authors Conclusion:
- “This is the first study that prospectively demonstrated the superior clinical efficacy of probiotics in patients with IBS with SIBO.”
- “The study also demonstrated that many clinical aspects mainly related with improvement of stool form and satisfaction from bowel function after probiotic treatment are seen among patients either with IBS-D or IBS-C, irrespective of the presence of SIBO.”
Limitations:
- This was a duodenal biopsy, not a jejunal biopsy. Theoretically, a jejunal biopsy is likely more accurate, although I don’t know until that is proven.
- This was an open-label study and had a small sample size.
Clinical Takeaways:
- Probiotics may work better for those whose IBS is driven by SIBO, inflammation or both.
- Probiotics can benefit IBS irrespective of SIBO
Dr. Ruscio Comments
It has been my longstanding belief that much of the benefit we see probiotic supplementation produce in IBS patients is via their antimicrobial action. It is disconcerting that some SIBO authorities recommend against the use of probiotics because ‘SIBO is a condition of overgrowth’. That is a theory, however, we have the clinical outcome data showing probiotics benefit IBS and that they are directly antibacterial. This study, albeit small, further strengthens this argument. Hopefully, with time, opinion will start to line up with the data. Also, it’s important to mention that occasionally someone will have a reaction probiotics, and if so simply discontinue therapy.
25-Hydroxyvitamin-D3 Serum Modulation After Use of Sunbeds Compliant with European Union Standards: A Randomized Open Observational Controlled Trial
https://www.ncbi.nlm.nih.gov/pubmed/28416344
Study purpose
- Asses the of impact of sunbed use compliant with the European Union standard on serum vitamin D [25 (OH) D] modulation and well-being
Intervention:
- In a randomized controlled study, 25(OH)D serum levels were measured at enrollment, after 1 week, and after completion of the 12-week period. Using sunbed twice weekly and compared with the control group without any sunbed exposure. 31 treatment, 23 controls.
- Sunbeds used: Lightech lamp Vita D (GE Lighting, East Cleveland, OH)
- Dose: One minimal erythema dose (enough to cause a slight pinkening of the skin post exposure)
- Duration of exposure per session was limited to a maximum of 20 minutes
Main Results:
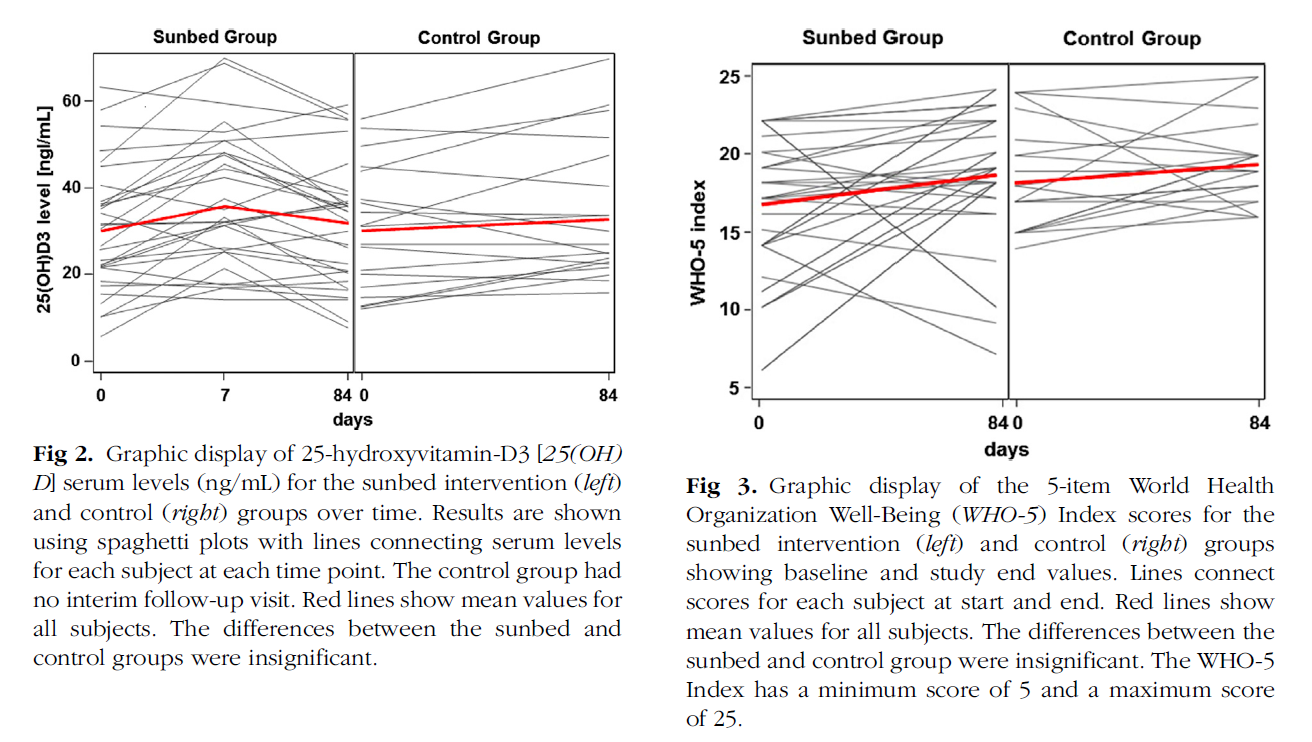
- After 1 week: sunbed intervention group (N = 31), had a 27% increase of mean 25(OH)D
- After 12 weeks, mean 25(OH)D levels had declined and were no longer different from baseline.
- After 12 weeks, 25(OH)D levels did not differ between the intervention and control group.
- Well-Being Index score did not differ between the sunbed and control groups
- Ironically, the control group (the group not exposed to tanning) actually experienced a significant increase in vitamin D
- The control group showed a statistically insignificant 11% (3.9 6 2.11 ng/mL) increase in mean vitamin-D serum levels over the 12-week period
Additional Results:
- It appears there are many unanswered questions regarding ideal UVA and UVB composition of tanning beds.
- UV phototherapy has been shown to increase serum 25(OH)D concentrations in several studies.7-9 However, almost all of the studies were performed with UVB broadband; only 1 study by Rogers et al investigated the vitamin-D plasma increase under psoralen plus UVA therapy
- Although the action spectrum for the conversion of 7-dehydrocholesterol to vitamin D3 is thought to be within the UVB range (280-310 nm), the results of Rogers et al suggested that the higher wavelengths of UVA (310-400 nm) may also have an effect, at least in the presence of psoralen.
- A more recent investigation found that narrowband UVB increases 25(OH)D values as early as 1 week after the start of phototherapy and consistently at week 12, whereas UVA1 resulted in a small but measurable decrease in 25(OH)D levels.
- Several studies have also reported an increase of 25(OH)D serum levels after sunbed exposure.5,12-16 Sunbeds typically emit UVA1 and UVA2 with small amounts of UVB However, in these investigations the follow-up was limited to at most 10 weeks
- This study used tanning beds compliant with European Union (EU) standard EN-60335-22-7. It is unclear if following these guidelines diminished the effect on vitamin D.
- The recent European Union (EU) standard EN-60335-22-7 recommended that the total effective irradiance must not exceed 0.3 W/m2, weighted according to the erythema action spectrum.
- It is therefore unclear to what extent the use of a sunbed consistent with these standards will influence serum levels of 25(OH)D.
- “Several investigations clearly showed an association between regular sunbed use and different cutaneous cancers. However, several investigators recently questioned the degree to which findings represent causation rather than association. They posit that the benefit of increased vitamin-D conversion after regular sunbed use may outweigh potential risks.”
Authors Conclusion:
- “In summary, our study showed only a transient impact of sunbed use on vitamin-D levels. We conclude that sunbed use compliant with the new EU standards does not contribute to a sustainable increase of vitamin-D serum levels nor does it improve self-reported well-being.”
Interesting Notes:
- Sun exposure may produce slightly more vitamin D than supplementing with 1,000 IUs per day
- “Several studies directly compared sunbed-related increase with the effects of daily 25(OH)D supplementation of 1000 IU per day, finding slightly higher 25(OH)D levels in the sunbed group compared with the oral supplementation group. However, follow-up was limited to a maximum of 8 to 10 weeks.”
Clinical Takeaways:
- Sunbeds compliant with the European Union standards may not lead to increased vitamin D.
Dr. Ruscio Comments
As I understand it, the EU’s standard for tanning beds are supposed to reduce risk and increase the safety of tanning beds. This is a laudable goal, however it appears that the guidelines may not allow adequate stimulation of the skin to lead to increased vitamin D product beyond a couple weeks. It may be the limitations on intensity of light or the amount of UVA and UVB used, the later details are unclear based upon this study. Michael Holick’s group published a critical appraisal of this paper which I am reading now and will include in next month’s edition.
Until proven safe, I would not recommend regular use of tanning beds. Rather I would recommend obtaining regular sun exposure during the sunny months and supplementing with vitamin D. There is also the DMinder app which Dr. Holick will be discussing in a future podcast. It helps you track the best time to be in the sun and how much vitamin D will be produced given your location.
Rapid-Fire Research – Ultra Concise Summaries of Noteworthy Studies
*please let me know how you like this very short summary format*
Follow-Up of Patients with Functional Bowel Symptoms Treated with a Low FODMAP Diet
https://www.ncbi.nlm.nih.gov/pubmed/27099444
- Low FODMAP was shown to be safe and efficacious for IBS and IBD in 16 months follow up study
- Most patients used modified low FODMAP diet, based upon reintroduction, as their long-term application of the diet
- One hundred and eighty patients were included, 131 (73%) IBS and 49 (27%) IBD patients.
- Median follow-up time was 16 mo (range: 2-80).
- Eighty-six percent reported either partial (54%) or full (32%) efficacy with greatest improvement of bloating (82%) and abdominal pain (71%).
- Satisfaction with dietary management was seen in 83 (70%) IBS patients and 24 (55%) IBD patients.
- Eighty-four percent of patients lived on a modified LFD, where some foods rich in FODMAPs were reintroduced, and 16% followed the LFD by the book without deviations.
- Wheat, dairy products, and onions were the foods most often not reintroduced by patients.
Aberrant Motility in Unaffected Small Bowel is Linked to Inflammatory Burden and Patient Symptoms in Crohn’s Disease
https://www.ncbi.nlm.nih.gov/pubmed/26509756
- Inflammation is a major driver of motility in Crohn’s disease patients
- Global SB (small bowel) motility was correlated with calprotectin, HBI, and symptom components (well-being, pain, and diarrhea).
- Even after adjusting for age, sex, smoking, and surgical history
- The associations remained highly significant after adjusting for covariates.
- Conclusion: “…inflammation-related enteric dysmotility may explain refractory abdominal symptoms in CD.”
- Notes: I feel the autoimmunity driven dysmotility in IBS and SIBO to have received more attention than it may deserve. Yes, it is a proven underlying cause, however, I am concerned that its importance has been over-reported. It is my hunch, albeit speculatively, that by improving one’s symptoms’, and thus reducing inflammation, we can allow the motility apparatus to repair. This study, although in Crohn’s disease, partially supports my contention.
A Proof-of-Concept Study Showing Antibiotics to be More Effective in Irritable Bowel Syndrome With than Without Small Intestinal Bacterial Overgrowth: A Randomized, Double-blind, Placebo-Controlled Trial
https://www.ncbi.nlm.nih.gov/pubmed/26731696
- 80 IBS patients (Rome III) were evaluated for SIBO by gut aspirate culture.
- Patients with and without SIBO were randomly assigned either antibiotic treatment, 800 mg/day norfloxacin for 10 days or placebo.
- Global symptom score (blindly), Rome III criteria, aspirate culture, and glucose hydrogen breath test (GHBT) were assessed before and 1 month after treatment, and patients were followed up for 6 months.
- RESULTS: Antibiotics, norfloxacin, was more effective than placebo at reducing the symptom score at 1 month among patients with SIBO. However, the scores were comparable at 6 months.
- CONCLUSION: Norfloxacin is superior to placebo in relieving symptoms of SIBO-associated IBS.
- Note: this study shows that antimicrobial therapy seems to work better in those with IBS + SIBO. The results here were short-lived, as compared to the results we discussed in the probiotic study above. Perhaps probiotics will be shown to have a better long-term effect than antibiotics in future comparative studies?
Prevalence of Thyroid Dysfunction and Autoimmunity in the Older Population and Implications of Age-Specific Reference Ranges
https://www.ncbi.nlm.nih.gov/pubmed/27988318
- The prevalence of SCH was higher in individuals ≥60 years, compared to that in individuals <60 years, but when age-specific TSH reference ranges were used, it was comparable between both study groups. In order to avoid misclassification in older individuals, it is important to use age-specific reference ranges in daily clinical practice.
- Note: be cautious not to jump to treatment when seeing elevated TSH in those over 60.
Subclinical Thyroid Dysfunction and the Risk of Cognitive Decline: A Meta-Analysis of Prospective Cohort Studies
https://www.ncbi.nlm.nih.gov/pubmed/27689250
- Eleven prospective cohorts followed 16,805 participants during a median follow up of 44.4 months.
- CONCLUSIONS: SHyper might be associated with an elevated risk for dementia, whereas SHypo is not, and both conditions are not associated with faster decline in MMSE over time. Available data are limited, and additional large, high-quality studies are needed.
- In random-effects models, the pooled adjusted risk ratio for dementia in SHyper was 1.67 (95% confidence interval, 1.04; 2.69) and 1.14 (95% confidence interval, 0.84; 1.55) in SHypo vs euthyroidism, both without evidence of significant heterogeneity (I2 = 0.0%).
- SHyper = SC Hyperthyroid. SHypo = SC Hypothyroid.
- Note: this study reinforces the lack of concern associated with subclinical hypothyroidism in aging populations. It does suggest that subclinical hyperthyroidism (TSH 0.1-0.4 mIU/L) might be detrimental.
Factors Affecting Gastrointestinal Absorption of Levothyroxine: A Review
https://www.ncbi.nlm.nih.gov/pubmed/28153426
- Many gastrointestinal disorders, such as celiac disease, atrophic gastritis, lactose intolerance, and Helicobacter pylori infection, may impede the absorption of levothyroxine. During treatment of these disorders, it is necessary to monitor serum thyroid-stimulating hormone and free T4 values to reduce the risk of developing iatrogenic hyperthyroidism.
- Soybeans and coffee have the greatest impact on the reduction of absorption, whereas vitamin C has the ability to increase it. Conversely, the effect of dietary fiber on the absorption of LT4 is not yet fully understood; further research is needed on this topic.
- Certain drugs and vitamins may decrease absorption
- A decrease in the absorption of LT4 is established and clinically significant when administered concomitantly with cholestyramine, colesevelam, lanthanum, calcium carbonate, calcium citrate, calcium acetate, iron sulfate, ciprofloxacin, aluminum hydroxide, sevelamer, or proton pump inhibitors.
- The majority of the interactions are clinically significant. They are based on the LT4 adsorption on interfering substances in the digestive tract, as well as a consequently reduced amount of the drug available for absorption. These interactions can be avoided by separating the administration of LT4 and the interfering substance.
- Note: I have seen many cases who needed a decreased dose of thyroid medication after improving their gut health. Their ‘elusive hypothyroidism symptoms’ also disappeared.
I’d like to hear your thoughts or questions regarding any of the above information. Please leave comments or questions below – it might become our next practitioner question of the month.

Like what your reading?
Please share this with a colleague and help us improve functional medicine
Discussion
I care about answering your questions and sharing my knowledge with you. Leave a comment or connect with me on social media asking any health question you may have and I just might incorporate it into our next listener questions podcast episode just for you!