Practitioner Research Review – March 2018
Dr. Michael Ruscio’s Monthly – Future of Functional Medicine Review Clinical Newsletter
Practical Solutions for Practitioners
In Today’s Issue
Research
- Leptin in Autoimmune Diseases
- Clinical Trial: the Combination of Rifaximin With Partially Hydrolysed Guar Gum Is More Effective Than Rifaximin Alone in Eradicating Small Intestinal Bacterial Overgrowth
- Autoimmunity Affects Health-Related Quality of Life in Patients With Hashimoto’s Thyroiditis
- Bacillus as Potential Probiotics: Status, Concerns, and Future Perspectives
![]()
Research
*Please note: the case study and research studies are not meant to be mutually reinforcing. There is often concept overlap, however the research studies are a collection of the most clinically meaningful research that has been published recently.
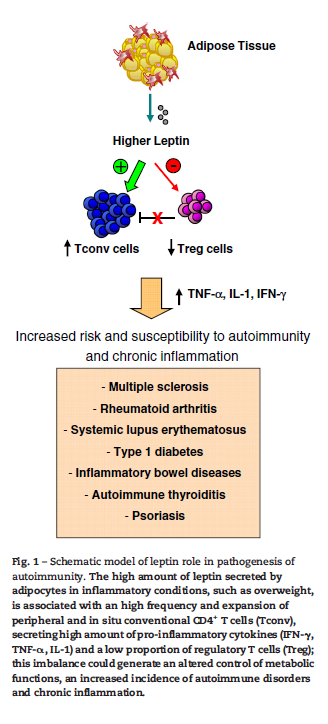
Leptin in autoimmune diseases.
https://www.ncbi.nlm.nih.gov/pubmed/25467840
Study purpose
- “Provide an overview of recent advances on the role of leptin in the pathogenesis of several autoimmune disorders.”
Intervention:
- Review, non-systematic.
Main Results:
- Leptin is pro-inflammatory
- The overall leptin action in the immune system is a pro-inflammatory effect, activating pro-inflammatory cells, promoting Th1 responses, and mediating the production of the other pro-inflammatory cytokines, such as TNF-α, IL-2, or IL-6. Leptin is, therefore, able to modulate both innate and adaptive immune response
- Higher leptin levels have been associated with autoimmunity
- Moreover, several studies in human revealed that leptin levels associated with autoimmune disorders, infections, and endocrine/metabolic diseases, thus suggesting a central role of leptin in immune homeostasis and in the pathogenesis of several inflammatory disorders
- Higher leptin associated with multiple sclerosis
- Indeed, two large studies, one in American women [48] and the other based on a Swedish population [49], demonstrated a twofold increased risk of developing MS among subjects with a BMI ≥30 kg/m2 at age 18 and 20 respectively, compared with normal weight subjects. Also, Munger et al. [50] found that a higher BMI at ages 7–13 was associated with a significantly increased risk of MS only among girls.
- Higher leptin associated with inflammatory bowel disease, and may directly drive inflammation in the intestines. However, some studies have not shown this association.
- The correlation between obesity and an increased risk of CD has been recently found in a large study
- Two clinical studies have correlated a high BMI with an unfavorable course of IBD, including a higher risk of relapses, abscesses, surgical complications … Leptin, in turn, induced epithelial wall damage and neutrophil infiltration that represent characteristic histological findings in acute intestinal inflammation
- High leptin can decrease regulatory T cells
- Fasting may benefit autoimmune disease via its ability to lower leptin, thus improving disease activity
- In addition, it has been reported that fasting, which is associated with a marked decrease in serum leptin concentration, determines a shift toward Th2-type cytokine secretion, thus improving clinical disease activity in RA patients by decreasing CD4+ activation. (1)
- Some clinical data has shown improved autoimmunity after fasting, namely in RA
- Finally, reduction of leptin levels in RA patients, by fasting, has been found to ameliorate the clinical symptoms of several autoimmune diseases including RA [13], thus suggesting that leptin antagonism could be proposed for the prevention of developing RA. (2)
- For thyroid autoimmunity, it appears thyroid hormone replacement can also decrease leptin, irrespective of changes in body weight. Note: other studies have shown thyroid hormone replacement can decrease thyroid antibodies.
- Treatment with T4 decreases leptin levels [191], without affecting BMI, strongly indicates an association between leptin and thyroid status.
- A recent cross-sectional study has reported a greater prevalence of hypothyroidism and HT-related autoantibodies among obese individuals, correlated with increased leptin levels.
- Another study [193] reported on a huge cohort study that childhood weight gain and childhood overweightness conferred a slightly increased risk of HT development at the age of 60–64 years, particularly in women
- Fasting can reduce inflammation
- Recent studies from Fontana et al.[197] have shown that caloric restriction and consequent lowering of serum leptin are able in humans to significantly reduce inflammatory parameters
Additional Results:
- NA
Authors Conclusion:
- “Leptin acts as a pro-inflammatory cytokine that promotes Th1 responses on one side and inhibits Treg cell expansion.:
Interesting Notes:
- na
Clinical Takeaways:
- Leptin may play a causal role in autoimmune diseases.
- Interventions that aid in weight loss, namely fasting, may help reduce inflammation, leptin and disease activity.
Dr. Ruscio Comments
Fasting is something to consider in patients with autoimmunity. Fasting likely has benefits for autoimmunity that go beyond leptin, most importantly improved gut health via less antigen presentation and providing a chance for the gut to rest and heal.
Be careful with fasting/intermittent fasting if someone is underweight and or does not react well to fasting (fatigue, irritability, brain fog, etc…). This can usually be picked up in your history, or fairly easily with some experimentation.
Clinical trial: the combination of rifaximin with partially hydrolysed guar gum is more effective than rifaximin alone in eradicating small intestinal bacterial overgrowth.
https://www.ncbi.nlm.nih.gov/pubmed/20937045
Study purpose
- To assess if the addition of partially hydrolyzed guar gum would enhance the effect of Rifaximin in treating SIBO.
Intervention:
- SIBO breath test positive patients were randomized to receive 10 days of
- rifaximin 1200 mg ⁄ day, or
- rifaximin 1200 mg ⁄ day plus partially hydrolyzed guar gum 5g⁄ day
- Note: a dose of 3.5-5 grams per day may be the ideal dose to achieve benefit while also minimizing the risk of reactions.
Main Results:
- 77 patients had SIBO
- Eradication rate of SIBO was:
- 62.1% in the rifaximin group
- 87% of the rifaximin plus partially hydrolyzed guar gum group
- Overall clinical improvement was observed in (for those who cleared SIBO + those who did not)
- 57% rifaximin
- 80% in rifaximin plus partially hydrolyzed guar gum
- Clinical improvement rates were higher in those who eradicated SIBO
- 87% rifaximin group and
- 91% rifaximin plus partially hydrolyzed guar gum groups respectively
Additional Results:
- Gradually increasing the dose of rifaximin might be better than using one level dose
- Recently, studies on SIBO diagnosed by GBT and treated with rifaximin reported that the progressive increase in rifaximin dosage (from 600 mg ⁄ day up to 1600 mg ⁄ day for 7 days) may lead to an important gain in terms of SIBO eradication without enhancing the occurrence of side effects.
Authors Conclusion:
- “Our results show that the combination of PHGG and rifaximin was significantly more effective than rifaximin alone in the eradication of SIBO. Thus, this association reached an eradication rate of 87.1%, which was significantly higher than that of rifaximin alone (62.1%).”
Interesting Notes:
- “Several studies have shown a beneficial effect on intestinal motility by use of alimentary fibres. In fact, alimentary fibres increase fecal mass and transit time, easing defecation”
Clinical Takeaways:
- The addition of prebiotics or fiber to SIBO antibiotic therapy may enhance SIBO clearance and symptomatic improvement
Dr. Ruscio Comments
This is a very interesting study and it suggests that adding prebiotics or fiber to your SIBO treatment might enhance results. This is especially true if using a prebiotic dose of 3.5-5 grams. This dose has the lowest chance of reactions but still will vector benefit.
That being said I have not made this a routine practice at my office. I am open but remain unconvinced that this is essential to adopt for all patients. Looking at the IBS literature and SIBO treatment literature collectively, it appears that the better evidence supports using probiotics. Prebiotics, in my opinion, are something to consider if optimal treatment results have not been obtained after the first round of treatment – similar to anti-biofilm agents.
Autoimmunity affects health-related quality of life in patients with Hashimoto’s thyroiditis.
https://www.ncbi.nlm.nih.gov/pubmed/27523457
Study purpose
- “aimed to reveal the effects of autoimmunity on health-related quality of life of euthyroid HT patients”
Intervention:
- 84 patients completed the SF-36 questionnaire. Note: This survey assesses various aspects (domains) that contribute to the quality of life (QOL).
- This includes patients who were on thyroid hormone Rx (62%) and patients who did not require meds. All patients had their hormone levels within normal lab ranges.
- Nearly, 62% of the patients were under thyroid hormone supplementation, in both the antibody negative and positive groups.
Main Results:
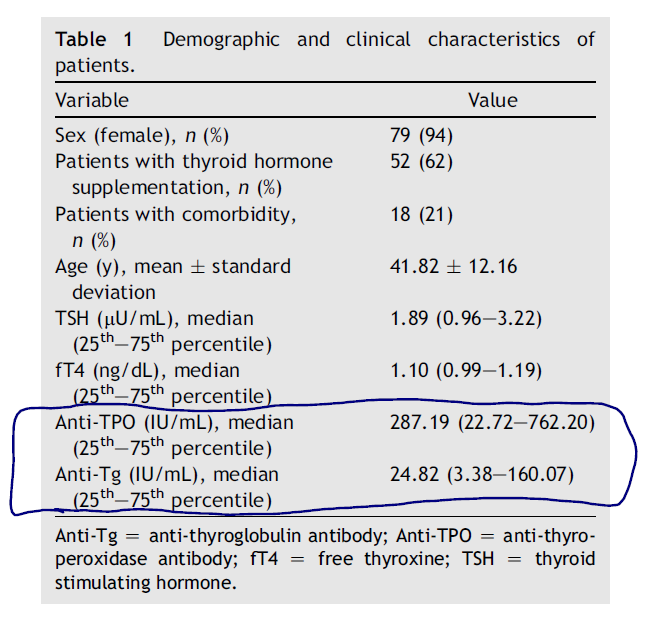
- There was an inverse relationship between antibody levels and quality of life scores
- There was a significant negative correlation between each domain score and the antibody levels, individually. Patients who had higher anti-TPO and anti-Tg levels had significantly lower quality of life domain score
- These results were maintained even after adjusting for co-morbidities
- When these groups were stratified by adjusting the comorbidities as a confounding factor; there was also a significant difference between both anti-TPO and anti-Tg antibody positive and negative groups
- There was no associated correlation with antibody levels and thyroid hormone levels
- There was statistically no significant correlation between the antibody levels and thyroid function tests
Additional Results:
- The TPO levels averaged 287
- This might be the first study to document a connection between TG antibodies and quality of life
- To the best of our knowledge, this is the first study demonstrating the negative correlation between anti-Tg and HRQoL
Authors Conclusion:
- “Our study revealed that higher thyroid antibody levels were negatively correlated with life quality scores.”
Interesting Notes:
- What causes the lower quality of life? The authors theorize it could be: selenium deficiency, thyroid hormone fluctuation, and disease awareness.
- If selenium deficiency is a cause of decreased QOL, then we should see improvement in the quality of life with selenium supplementation. While not all the data agree here, there is data to support 3-6 month of selenium use for both quality of life and thyroid antibodies (3)
- TPO is a more relevant clinical marker than TG, TG will be positive in a higher number of normal patients.
- Anti-TPO is considered to be the best serological marker for the diagnosis of HT and is found positive in nearly 95% of the patients
- Anti-Tg antibodies are positive in nearly 60-70% of HT patients and are found to be positive in a higher proportion of healthy people.
- Not all studies show thyroid autoimmunity is associated with depressed mood, namely depression (4)
Clinical Takeaways:
- Thyroid autoimmunity is associated with lower quality of life scores.
- This effect may be independent of thyroid hormone levels or use of thyroid replacement medication.
- Selenium supplementation, for 3-6 months, may improve health-related quality of life.
Dr. Ruscio Comments
Thyroid autoimmunity appears to affect the quality of life and might do so irrespective of thyroid hormone status. This is not surprising given that Hashimoto’s is an inflammatory process. What is important to keep in mind is what levels of antibodies are considered a ‘clinical win’. While we don’t have a definitive answer to this question, I look at TPO levels in the lower hundreds (100 – 400 roughly) as acceptable.
Also, remember that the TG antibodies are not always significant. I had to remind a patient of this just the other day. After a few months of care, including the use of selenium, CoQ10, magnesium and vitamin D for her antibodies, all of her chief complaints were markedly improved, however, she still had high levels of TG antibodies. She was very concerned about this until I explained that elevated TG antibodies can be ‘normal’ in some people and she need not worry.
Finally, remember that there is more to the quality of life than one’s antibody levels. Disease awareness is another. If you mismanage this conversation you could exacerbate someone’s fear and decrease their quality of life.
Bacillus As Potential Probiotics: Status, Concerns, and Future Perspectives
https://www.ncbi.nlm.nih.gov/pubmed/28848511
Study purpose
- Review utility and concerns regarding Bacillus, aka soil-based or spore-forming, probiotics.
Intervention:
- Review, non-systematic.
Main Results:
- Bacillus probiotics are heat, acid and bile stable
- Bacillus spp. can survive in extreme acidity of stomach and tolerate bile salts
- They also exert antimicrobial, antioxidant and immune-modulatory effects.
- Bacillus spp. have higher acid tolerance and better stability during heat processing and low-temperature storage. Additionally, they have also been shown to possess pathogen exclusion, anti-oxidant, antimicrobial, immuno-modulatory and food fermentation abilities.
- Bacillus have anti-biofilm activity
- Exclusion of pathogen by the inhibition of bacterial biofilm is another potential attribute proposed for Bacillus strains.
- They can also help with digestion of carbs, proteins, and fats
- Bacillus spp. have been used for the production of food grade amylase, glucoamylase, protease, pectinase and cellulase in varying foodstuffs
- Some Bacillus are pathogenic
- Few of the members of Bacillus spp. particularly, B. cereus, B. weihenstephanensis, B. anthracis, and B. thuringiensis species are known to produce various toxins, including ematic or enterotoxin (Cereulide), Bipartite exotoxins: protective antigen- lethal factor (PA-LF) and PA-edema factor (PA-EF), Cry and Cyt
- Bacillus is very similar to Lactobacillus
- The genus Bacillus is closely related to Lactobacillus spp., the distinguished candidate probiotic. Both share the same class, Bacilli under the phylum Firmicutes
- Bacillus are not a normal inhabitant of the human GI
- It is believed that Bacillus spp. are not a natural inhabitant of the gut. They get colonized into the intestinal tract after consumption of vegetables or raw food materials contaminated with soil.
- However, Bacillus do appear in the stool of the majority of humans
- A study by Tam et al. (2006) demonstrated that spores of Bacillus spp. could readily be recovered in the range of 10^3-10^8cfu/g of human feces.
- The 16S rRNA gene phylogenetic analysis of isolates demonstrated the presence of 10 different Bacillus species in examined fecal samples of 30 volunteers (Tam et al., 2006).
- The most studied strains are B. coagulans and B. subtilis
- B. clausii and B. licheniformis appear to be the most common strains found in human stool
- Hoyles et al. (2012) further studied the diversity of Bacillus spp. and related spore-former bacteria in human feces and documented that the majority of recovered isolates belonged to Bacillaceae family. Two species, B. clausii and B. licheniformis were recovered most frequently
- Adverse events after using Bacillus probiotics appears rare
- All appear to show no indications of adverse effects indicating the most occurrence of illness associated with Bacillus probiotic strains supplement of humans are result of either opportunistic infections or miss-diagnosis
- B. coagulans, subtilis, clausii and licheniformis appear common in food products
- Respectively, the strains of B. subtilis, B. subtilis var. natto, B. clausii, B. licheniformis, and B. coagulans etc. are being utilized to improve the quality and demand of functional foods globally
- Bacillus species have a transient impact, notably on the small intestine
- They stated that the spores germinated in significant numbers in the jejunum and ileum, suggesting their colonization into the small intestine. Recently, Ghelardi et al. (2015) documented that the orally administered Bacillus spp. follows transient colonization into the intestine
- Bacillus probiotics may be contraindicated for those with immunosuppression and/or those who are critically ill
- Although Bacillus probiotics have an overall excellent health-promoting record, especially in preventing and curing of diarrhea, gingivitis, H. pylori infection and maintaining homeostasis of the intestine (Khodadad et al., 2013; Lefevre et al., 2015; Alkaya et al., 2016; Lopetuso et al., 2016). Their application with certain immune deficient population especially for critically ill, neonates and elderly groups should be evaluated and regulated carefully since reports related to bacteremia in immune-compromised patient treated with spore former and other probiotics have been recorded repetitively
Additional Results:
- Like all probiotics, using a reputable brand is important – mostly to ensure the label claims are met.
- Likewise, the entitlements of probiotic products being marketed as functional food, dietary supplement or drug are also elucidated by the state of probiotic strains. Thereby, the quality of commercial probiotic products is an important issue to be considered for regulation. Several researchers have identified discrepancies between labeled and actual contents of commercial probiotic products
- Like other probiotics, Bacillus may positively impact other members of the microbiota
- After 28 day treatment of Bacillus coagulans in elderly subjects, baseline populations of Faecalibacterium prausnitzii, Clostridium lituseburense and Bacillus spp. were significantly higher, relative to the placebo group
- Some researchers feel that Bacillus should be considered healthy commensals
- In this regard, Hong et al. (2009) reinforced a growing view that B. subtilis and probably other species had adapted to life within the human GIT, including the ability to form a biofilm, sporulate anaerobically and produce antimicrobials, and should be considered gut commensals, rather than solely soil microorganisms.
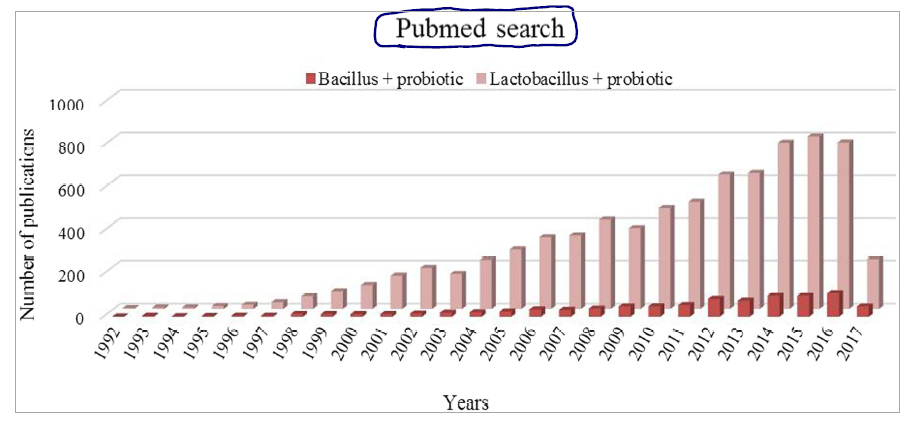
- There appears to be a favoring of more traditional probiotic strains, like Lactobacillus and Bifidobacterium species, in the published literature
Authors Conclusion:
- “Spore-forming bacilli are being explored for the production and preservation of food for many centuries. The inherent ability of production of a large number of secretory proteins, enzymes, antimicrobial compounds, vitamins, and carotenoids specifies the importance of bacilli in the food chain.”
Interesting Notes:
- Bacillus probiotics are rightly referred to as soil-based probiotics
- Bacillus signifies a Gram-positive, rod-shaped, spore-forming, aerobic or facultative anaerobic bacterium. In general, the genus Bacillus is designated as a group of soil inhabitants. However, Bacillus spp. can be isolated from varied sources including air, water, human and animal gut, and also from vegetables and food
- Fermented foods are a common source of Bacillus probiotics
- The consumption of vegetative cells and spores of Bacillus spp. by human beings is frequent through fermented foods and raw vegetables
- I organize probiotics into 4 categories (1. Lacto/Bifido, 2. S. boulardii, 3. Soil-based, 4. E. coli 1917), these authors suggest a similar model of 5 groups
- If we look at the types of organisms, the probiotic market is categorized into five groups, i.e., Lactobacillus, Bifidobacterium, spore formers, yeast, and others. Hence, the spore former probiotics are making a major contribution in global nutraceutical as well as the pharmaceutical market.
Clinical Takeaways:
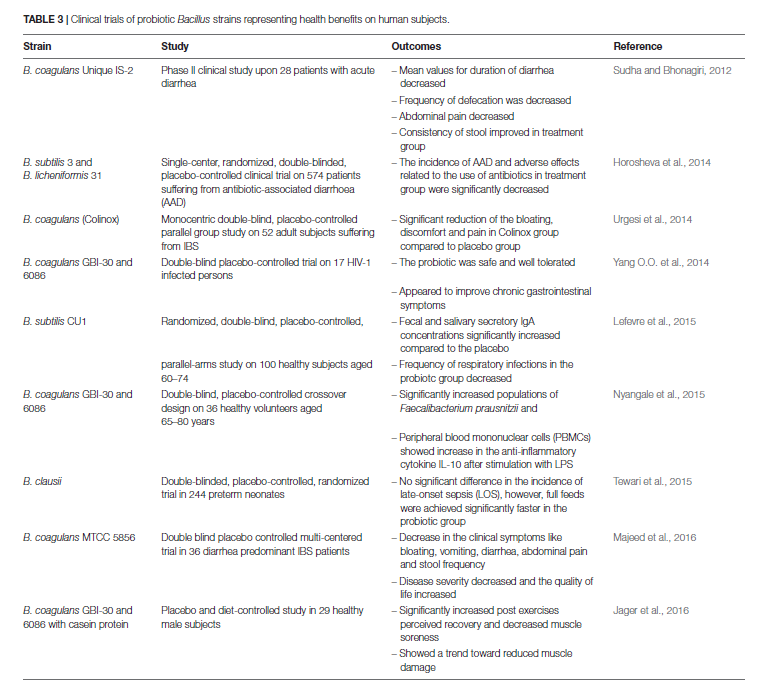
- Bacillus (aka spore-forming or soil-based) probiotics appear safe and to have some documented health benefits.
- B. subtilis and B. coagulans are the most well studied.
- Adverse events are rare but these should be avoided in those who are critically ill or immunosuppressed
- Most studies use an upper dose range of 2-8 billion CFU/day.
Dr. Ruscio Comments
The paleo community has been excited about Bacillus probiotics because of their philosophical congruity. However, it is important to evaluate how a theoretical posit stands up to scientific scrutiny. In this case, it appears that yes, Bacillus probiotics are both sound according to ancestral wisdom and this supposition has been supported by modern science.
Somewhat ironically, probiotics (including Bacillus) may produce their favorable impact not by ‘colonizing the host’ but by being directly antibacterial and antifungal.
Clinically, they are worth a trial and will likely benefit many patients. However, remember that there will always be those who react to a given therapy. Be on the lookout for this and consider other classes of probiotics, or avoid probiotics entirely, depending on the response of the individual.
I’d like to hear your thoughts or questions regarding any of the above information. Please leave comments or questions below – it might become our next practitioner question of the month.

Like what your reading?
Please share this with a colleague and help us improve functional medicine

Discussion
I care about answering your questions and sharing my knowledge with you. Leave a comment or connect with me on social media asking any health question you may have and I just might incorporate it into our next listener questions podcast episode just for you!